Research Article - Journal of Parasitic Diseases: Diagnosis and Therapy (2017) Volume 2, Issue 2
In vitro anticoccidial, antioxidant activities and cytotoxicity of Psidium guajava extracts.
Yamssi Cedric1*, Vincent Khan Payne1, Noumedem Anangmo Christelle Nadia1, Norbert Kodjio2, Etung Kollins1, Leonelle Megwi1, Jules-Roger Kuiate2, Mpoame Mbida1
1Research Unit of Biology and Applied Ecology, Faculty of Science University of Dschang, Cameroon
2Research Unit of Microbiology and Antimicrobial Substances, Faculty of Science, University of Dschang, Cameroon
- *Corresponding Author:
- Yamssi Cedric
Department of Biology and Applied Biology
Faculty of Science
University of Dschang
PO Box 067, Dschang, Cameroon
Tel: (237) 677365519
E-mail: cyamssi@yahoo.com
Accepted October 13, 2017
Citation: Cedric Y, Payne VK, Nadia NAC, et al. In vitro anticoccidial, antioxidant activities and cytotoxicity of Psidium guajava extracts. J Parasit Dis Diagn Ther. 2017;2(2):14-24
Abstract
Background: Coccidiosis remains one of the most important infectious causes of digestive disorders in rabbits. The aim of this study was to evaluate in vitro anticoccidial and antioxidant activities of Psidium guajava extracts. Methods: Sporulation inhibition bioassay was used to evaluate the activity of Psidium guajava extracts on sporulation of Eimeria flavescens, Eimeria stiedae, Eimeria intestinalis and Eimeria magna oocysts and sporozoites. The set up was examined after 24 h and 48 h for the oocysticidal activities and after 12 h and 24 h for anti-sporozoidal activities. The antioxidant activity was determined by measuring FRAP (ferric reducing-antioxidant power), 1,1-diphenyl-2- picrylhydrazyl (DPPH) free radical scavenging and nitric oxide (NO) radical scavenging. The cytotoxicity of the most active extract was determined against animal cell lines fibroblast L929, HEPG2 and Hella cells using MTT assay. The impact of the toxicity was established by analysing the Selectivity Index (SI) values. Results: The highest efficacy of tested plant extracts was recorded after 24 h, which varied according to different concentrations of the tested extracts. The highest efficacy was 88.67 ± 2.52% at the concentration of 30 mg/ml of the methanolic extract against E. intestinalis. Most extracts including the aqueous extract exhibited good anti-sporozoidal activities against E. flavescens, E. stiedae, E. intestinalis and E. magna sporozoites at 1000 µg/ml. The highest viability inhibitory percentage was 97.00 ± 1.73% at a concentration of 1000 µg/ml of P. guajava methanolic extract against E. intestinalis sporozoites. These results also showed that methanolic and Ethyl Acetate extract, possessed strong antioxidant activities (IC50<20 µg/ml). The methanolic extract of P. guajava exhibited CC50 of>30 µg/ml against selected cell lines, suggesting that the compounds are not toxic. Phytochemical screening of the most active extract showed presence of alkaloids, flavonoids, saponins and phenols. Conclusion: These results provide confirmation to the usage of Psidium guajava against coccidioses by Agricultural farmers in Cameroon.
Keywords
Psidium guajava, Anticoccidial activity, Antioxidant, Eimeria species, Cameroon.
Introduction
In recent years, there has been increasing commercial production of rabbits as a source of protein. The consumers prefer rabbits for their low cholesterol and fat contents and high levels of essential amino-acid [1]. In addition to this commercial value, these animals are used as very important models for medical research and as pets [2]. Therefore, rabbit production become one of the important animal resources in the world [1]. However, coccidiosis remains one of the most important infectious causes of digestive disorders in rabbits [3]. According to a recent estimate [4], coccidiosis may cost the US rabbit industry about $127 million annually and likewise similar losses may occur worldwide.
Coccidiosis is caused by intracellular protozoon parasites of the genus Eimeria and causes significant mortality in domestic rabbits. Coccidiosis is one of the most frequent and prevalent parasitic diseases, accompanied by weight loss, mild intermittent to severe diarrhea with faeces containing mucus or blood and results in dehydration, decreased rabbit breeding [5]. The disease is seen most often in rearing establishments where sanitation is poor. So far, 15 species of Eimeria in rabbits have been identified [6]. Today 14 species of Eimeria are known to infect the intestine while one is located in the biliary duct of the liver. Two types of coccidiosis, intestinal and hepatic are described in rabbits. The intestinal coccidial species which cause weight reduction, diarrhoea and mortality due to villi atrophy leads to malabsorption of nutrients, electrolyte imbalance, anaemia, hypoproteinemia and dehydration [7]. The rabbit intestinal coccidia parasitize distinct parts of the intestine and at different depths of the mucosa [8]. Thus coccidiosis is probably the most expensive and wide spread infectious disease in commercial rabbit systems.
Most of the current anti-coccidial drugs show low efficacy and cause deleterious side effects. The extensive use of chemical anti-coccidial drugs in controlling this disease has led to the development of drug-resistant parasites [9]. Parasite resistance and the side effects of some of the anti-coccidial drugs have serious consequences on disease control. In the surrounding environment, commonly used disinfectants include some phenolic products such as ammonia, methyl bromide and carbon disulfide. Toxic effects of these products represent a danger to the staff and health of animals and therefore their use has been restricted [10]. Because of widespread drug resistance constraints [11], residual effects of drugs in meat of animals and toxic effects of disinfectants, scientists all over the world are shifting towards alternative approaches for the control of parasitic problems [12].
In various physiological and pathological conditions, the systemic amount of free radicals and reactive oxygen species are higher than normal. Free radical oxidative species are known to be produced during the host’s cellular immune response to invasion by Eimeria species [13], which plays an important role in defending against parasitic infections.
Another free radical oxidative species, nitric oxide promotes vasodilation and hemorrhage in coccidian infections which could be toxic to both parasites as well as to host cells harboring the coccidian parasite [14].
Georgieva et al. [15] observed that E. acervulina oocytes motivate lipid peroxidation, increase oxidative damage and imbalance in the antioxidant status in infected animals by disturbing the oxidative balance. Therefore to alleviate or reduce the oxidative stress, natural (e.g. Vitamin E, Se) and synthetic (e.g. butylated hydroxytoluene) antioxidants as feed supplements are commonly used in the poultry industry.
The use of antioxidants as anticoccidial remedies, therefore, holds promise as an alternative in the control of coccidiosis. Today, the use of antioxidant- rich plant extracts has gained special importance because of restriction in the use of synthetic compounds against coccidial infections due to emergence of resistance and their drug residues [16]. Naidoo et al. [17] also described antioxidant rich plant extracts as potential candidates in controlling coccidiosis in poultry. Therefore, the use of natural antioxidants may alleviate difficulties related to synthetic drugs, as they are not only natural products but may comprise new molecules to which resistance has not yet developed.
Psidium guajava is a medicinal plant used in tropical and subtropical countries to treat many health disorders. It has been reported that Psidium guajava leaf extract has a wide spectrum of biological activities such as anticough, antibacterial, haemostasis [18,19], antidiarrhoeal narcotic [20], and antioxidant properties [21]. This work was therefore aimed at evaluating the anticoccidial and antioxidant activities of crude extracts of P. guajava in order to justify its usage by Agricultural farmers as an anticoccidial drug.
Materials and Methods
Plant material
The leaves of Psidium guajava were collected in Menoua Division, Western Region of Cameroon and identified by Mr. NGANSOP Eric, a botanist at the Cameroon National Herbarium (Yaoundé) using a voucher specimen registered under the Reference No 2884/SRF.
Preparation of extract
Methanol, hexane and Ethyl Acetate extracts were obtained using the procedure described by Wabo Poné et al. [22]. Briefly, 100 g of stored powder were macerated in 1.5 L of each of the organic solvents. This helped to remove the principal natural compounds of the plants [23]. The mixture was stirred daily and 72 h later, these solutions were then filtered using Whatman Paper N 3. The filtrate was concentrated by evaporating the solvent at 75°C using a rotatory evaporator (Buchi R-200) to obtain the extracts.
For the aqueous extract (Infusion), a similar procedure was carried out except for the fact that distilled water was heated at 100°C and 100 g of the stored powder were poured into 1.5 L of hot distilled water. The mixture was stirred and the solution filtered using a tea sieve and filter paper. The methanolic, hexane, Ethyl Acetate and aqueous extracts obtained were kept in a refrigerator at 4ºC for further processing.
Anticoccidial activities of the extracts
Preparation of culture media
Dichromate (K2Cr2O7) Potassium: 2.5% Potassium dichromate were prepared by dissolving 2.5 g of potassium dichromate in 100 ml of distilled water. This culture medium was stored and used to prepare our plant extract concentrations.
Preparation of hanks buffered salt solution (HBSS):
Buffer HBSS: KCl …………………….0.4 g
KH2PO4 ……………… .0.06 g
NaCl ……………………8.0 g
NaHCO3 ……….……….0.35 g
Na2HPO4 ……………….0.048 g
D-glucose ………………1.0 g
Water was added up to 1L and the buffer frozen for storage
Preparation of the excystation solution: 125 ml of HBSS were added to 0.32 g of trypsin, 0.25 g Bile Salt and 0.3 g of taurocholate and the pH was adjusted to 7.6 using NaOH.
Preparation of sporulated oocysts: Field Isolates of Eimeria flavescens oocysts were collected from the large intestine while occysts of E. stiedae were collected from the gall bladders and necrotic hepatic lesions of naturally infected rabbits. These oocysts were washed and concentrated by the flotation method [24]. The sporulated oocysts were stored in 2.5% potassium dichromate at 4°C until they were used for experimental infections. Eimeria intestinalis and Eimeria magna were kindly provided by Alisson Niepceron (INRA, BASE, Tours, France). The Eimeria flavescens, Eimeria intestinalis, Eimeria magna and E. stiedae field isolates were maintained by periodic passage through young Rabbits in the Laboratory of Biology and Applied Ecology.
Preparation of stock solutions: For the aqueous extracts, 1200 mg of each extract were weighed using an electric scale balance and then 20 ml of distilled water introduced into the mortar. After homogenization, the mixture was transferred into a beaker. For the organic extract, a stock solution was equally prepared and the same amount of dry extract was first mixed with 0.3 ml of Dimethyl sulfoxide (DMSO) to facilitate dissociation of the organic extract with water. Stock solutions with a concentration of 40 mg/ml were thus obtained. By successive dilutions, we obtained solutions of concentration 40, 20, 10 and 5 mg/ml for the oocysticidal evaluation. For the anti sporozoidal evaluation, a working stock solution of 2000 μg/ml of the plant extract solution was prepared by weighing 20 mg of crude extract and dissolving it in 10 ml of distilled water. This was well mixed and serial dilution was carried out to obtain solutions of concentration 1500, 1000, 500, 250 μg/ml.
In vitro oocysticidal effect of extracts: Petri dishes were used to evaluate in vitro disinfectant activities. Each well contained a total volume of 2 ml of each concentration of the extracts (2.5, 5, 10, 20 and 30 mg/ml) inoculated with equal number of unsporulated oocysts and incubated at 28°C. For comparison, phenol was used as the reference disinfectant. The set up was examined after 24 h and 48 h. The number of sporulated and non-sporulated oocysts were counted and the percentage of sporulation was estimated by counting the number of sporulated oocysts in a total of 100 oocysts. The sporulation inhibitory percentage was calculated as follows.
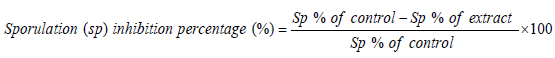
In vitro anti-sporozoidal effect of extracts: Stored oocysts in K2Cr2O7 were washed several times with HBSS (pH 7.2) until the K2Cr2O7 was completely removed. The oocysts were then incubated in a water bath at 41oC and shaken during incubation for 60 min. The suspension was centrifuged at 3,000 – 5,000 x g 10 min and resuspended in HBSS. Liberated sporozoites were washed with HBSS. The sporozoites were counted using the malassez counting chamber.
Petri dishes were used to evaluate the in vitro sporocidal activities. Each well contained a total volume of 2 ml of each concentration of the extracts (125, 250, 500, 750 and 1000 μg/ml) and inoculated with equal number of sporozoites. For comparison, amprocox was used as the reference drug. The set up was examined after 12 h and 24 h. The number of viable and non-viable sporozoites were counted and the percentage of viability was estimated by counting the number of viable sporozoites in a total of 100 sporozoites.
The viability inhibitory percentage was calculated as follows.
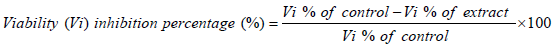
Antioxidant activities
The 2,2-Diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay: The radical scavenging activities of crude extracts were evaluated spectrophotometrically using the 1,1-diphenyl-2- picrylhydrazyl (DPPH) free radical [25]. When DPPH reacts with an antioxidant compound which can donate hydrogen, it is reduced. The changes in color were measured at 517 nm under UV/Visible light spectrophotometer (Jenway, Model 1605). Pure methanol was used to calibrate the counter. The extract (2000 μg/mL) was twofold serially diluted with methanol. One hundred microliters of the diluted extract were mixed with 900 μL of 0.3 mM 2,2-diphenyl-1-picrylhydrazyl (DPPH) methanol solution, to give a final extract concentration range of 12.5 - 200 μg/mL (12.5, 25, 50, 100 and 200 μg/mL). After 30 min of incubation in the dark at room temperature, the optical densities were measured at 517 nm. Ascorbic acid (Vitamin C) was used as control. Each assay was done in triplicate and the results, recorded as the mean ± standard deviation (SD) of the three findings, were presented in tabular form. The radical scavenging activity (RSA, in %) was calculated as follows:
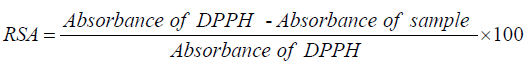
The radical scavenging percentages were plotted against the logarithmic values of concentration of test samples and a linear regression curve was established in order to calculate the RSA50 or IC50 which is the concentration of the sample necessary to decrease by 50% the total free DPPH radical [26].
Ferric reducing/antioxidant power (FRAP) assay: The ferric reducing power was determined by the Fe3+ - Fe2+ transformation in the presence of the extracts. The Fe2+ was monitored by measuring the formation of Perl’s Prussian blue at 700 nm. Different volumes (400, 200, 100, 50, 25 μL) of methanolic extracts prepared at 2090 μg/mL were mixed with 500 μL of phosphate buffer (pH 6.6) and 500 μL of 1% potassium ferricyanide and incubated at 50°C for 20 min. Then 500 μL of 10% trichloroacetic acid was added to the mixture and centrifuged at 3000 rpm for 10 min. The supernatant (500 μL) was diluted with 500 μL of water and mixed with 100 μL of freshly prepared 0.1% ferric chloride. The absorbance was measured at 700 nm. All the tests were performed in triplicate and the results were the average of three observations. Vitamin C was used as a positive control. Increased absorbance of the reaction mixture indicated a higher reduction capacity of the sample [27].
Nitric oxide radical scavenging (NO) assay: The method reported by Chanda and Dave [28] was used with slight modification. To 0.75 mL of 10 mM sodium nitroprusside in phosphate buffer was added 0.5 mL of extract or reference compounds (Vitamin C and Butylated hydroxytoluene (BHT)) in different concentrations (62.5 - 1000 μg/mL). The resulting solutions were then incubated at 25°C for 60 min. A similar procedure was repeated with methanol as blank which served as negative control. To 1.25 mL of the incubated sample 1.25 mL of Griess reagent (1% sulfanilamide in 5% phosphoric acid and 0.1% N-1-napthylethylenediamine dihydrochloride in water) were added. A final concentration range of 12.5 - 200 μg/ mL (12.5, 25, 50, 100 and 200 μg/mL) was obtained. After 5 min of incubation in the dark at room temperature, absorbance of the chromophore formed was measured at 540 nm. Percent inhibition of the nitrite oxide generated was measured by comparing the absorbance values of control and test samples. The percentage of inhibition was calculated according to the following equation:
Where, Al=absorbance of the extract or standard and A0=absorbance of the negative control.
Total phenol contents (TPC): The amount of total phenols was determined by Folin-Ciocateu Reagent method. The reaction mixture consisted of 20 μL of extract (2000 μg/mL), 1380 μL of distilled water, 200 μl of 2N FCR (Folin Ciocalteu Reagent) and 400 μL of a 20% sodium carbonate solution. The mixture was incubated at 40°C for 20 min. After cooling, the absorbance was measured at 760 nm. In the control tube, the extract volume was replaced by distilled water. A standard curve was plotted using Gallic acid (0-0.2 μg/mL). The tests were performed in triplicate and the results expressed as milligrams of Gallic Acid Equivalents (mgGAE) per gram of extract.
Total flavonoid content (TFC): The amount of total flavonoids was determined by the Aluminum chloride method. Methanolic solution of extracts (100 μL, 2000 μg/ml) was mixed with 1.49 mL of distilled water and 30 μL of a 5% NaNO2 solution. After 5 min, 30 μL of 10% AlCl3H2O solution were added. After 6 min, 200 μl of 0.1 M sodium hydroxide and 240 μl of distilled water were added. The solution was well mixed and the increase in absorbance was measured at 510 nm using a UV-Visible spectrophotometer. Total flavonoid content was calculated using the standard catechin calibration curve. The results were expressed as milligrams of Catechin Equivalents (mgCE) per gram of extract.
Evaluation of plant extracts cytotoxicity
The cytotoxicity of the most active extract was evaluated on animal cell lines fibroblast L929, HEPG2 and Hella cells using MTT assay as described by Mosmann [29]. Briefly, cells (104 cells/200 ml/well) were seeded into 96-well flat-bottom tissue culture plates in complete medium (10% foetal bovine serum, 0.21% Sodium Bicarbonate (Sigma, USA) and 50 mg/ml gentamicin). After 24 h, plant extracts at different concentrations were added and plates incubated for 48 h in a humidified atmosphere at 37ºC and 5% CO2. 10% DMSO (v/v) was used as a positive inhibitor. Thereafter, 20 μl of a stock solution of MTT (5 mg/mL in 1X phosphate buffered saline) were added to each well, gently mixed and each plate incubated for another 4 h. After spinning the plates at 1500 rpm for 5 min, supernatants were removed and 100 ml of 10% DMSO were added in each well to stop the reaction of extracts. Formation of formazon obtained after transformation of tetrazolium was read on a microtiter plate reader at 570 nm. The 50% cytotoxic concentration (CC50) of plant extract was determined by analysis of dose–response curves, according to the cytotoxicity gradient of plant extracts established by Malebo et al. [30]. Also, the Selectivity Index (SI) was calculated using the following formula:
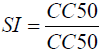
Phytochemical screening
The most active extract was tested for the presence of phenolic compounds, alkaloids, flavonoids, Polyphenols, tannins, saponin, triterpenes and steroids using standard procedures described by Builders et al. [31].
Statistical analysis
The data obtained were analyzed using one-way analysis of variance (ANOVA) and presented as mean ± standard deviation (SD) of three replications. The levels of significance, considered at P<0.05, were determined by Waller-Duncan test using the Statistical Package for Social Sciences (SPSS) software version 12.0.
Results and Discussion
Results
Anticoccidial activities
In vitro oocysticidal activities of P. guajava extracts: The in vitro oocysticidal activity of different extracts from the plants against Eimeria intestinalis, Eimeria magna, Eimeria flavescens and Eimeria stedai strains is summarized in (Table 1). It can be seen from Table 1 that about 90% of oocysts of Eimeria sp managed to sporulate in the control incubations containing oocysts and DMSO or K2Cr2O7. The highest efficacy of tested plant extracts was recorded after 24 h. post exposure which varied according to different concentrations of the tested extracts. Concerning P. guajava extracts, the highest efficacy was 88.67 ± 2.52% at the concentration of 30 mg/ml of methanolic extracts against Eimeria intestinalis. On the contrary the lowest efficacy was 7.00 ± 4.36% at the concentration of 2.5 mg/ml of the hot water extract on Eimeria flavescens after 48 h of incubation. Passing through the other used concentrations of P. guajava extracts (2.5, 5, 10 and 20 mg/ ml), they showed reduced efficacy depending on species of Eimeria tested.
| Conc mg/ml | Extract | Incubation time and Eimeria strains | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | ||||||||
| E. intestinalis | E. magna | E.flavescens | E. stedai | E. intestinalis | E. magna | E. flavescens | E. stedai | ||
| 2.5 | IF | 17.00 ± 11.53ab | 9.00 ± 2.65a | 7.67 ± 3.06a | 19.67 ± 3.10a | 9.00 ± 5,57a | 8.00 ± 3.61a | 7.00 ± 4.36a | 16.00 ± 1.73a |
| HE | 13.33 ± 1.16a | 18.00 ± 3.00b | 10.33 ± 2.08b | 11.00 ± 2.65ab | 12.33 ± 1.16a | 16.67 ± 1.53b | 9.67 ± 1.53a | 7.00 ± 3.61a | |
| EA | 21.67 ± 2.52ab | 20.00 ± 2.00b | 27.00 ± 6.56c | 21.33 ± 1.53ab | 20.33 ± 3.06b | 18.67 ± 2.52b | 25.67 ± 6.03b | 18.33 ± 3.06a | |
| ME | 31.33 ± 4.16b | 21.67 ± 1.53b | 27.00 ± 6.56d | 23.67 ± 1.53b | 23.67 ± 2.89b | 20.00 ± 1.00b | 25.67 ± 6.03b | 19.00 ± 1.00a | |
| 5 | IF | 15.00 ± 1.00a | 11.67 ± 2.52a | 13.33 ± 1.53a | 25.33 ± 3.22a | 12.33 ± 1.53a | 10.00 ± 3.00a | 12.00 ± 1.00a | 21.00 ± 2.65a |
| HE | 36.67 ± 3.22b | 23.33 ± 3.21b | 31.00 ± 6.56b | 13.67 ± 2.52b | 34.67 ± 3.22b | 22.00 ± 2.65b | 29.33 ± 6.66b | 9.00 ± 3.00b | |
| EA | 38.33 ± 4.04b | 28.33 ± 2.08b | 48.33 ± 6.35b | 30.33 ± 2.08c | 37.33 ± 3.51b | 26.33 ± 3.06b | 47.67 ± 6.66b | 25.33 ± 3.06b | |
| ME | 53.33 ± 6.11c | 38.33 ± 5.51c | 48.33 ± 6.35c | 39.67 ± 4.51d | 47.67 ± 8.51c | 36.67 ± 5.51c | 47.67 ± 6.66c | 36.33 ± 6.51c | |
| 10 | IF | 38.00 ± 4.00a | 26.00 ± 4.00a | 31.00 ± 2.00a | 38.00 ± 4.36a | 35.67 ± 4.51a | 24.33 ± 4.04a | 29.33 ± 2.08a | 34.67 ± 4.04a |
| HE | 47.00 ± 4.58b | 36.33 ± 4.16b | 40.67 ± 1.53b | 27.33 ± 3.06b | 45.67 ± 4.51b | 35.00 ± 4.58ab | 39.33 ± 2.08b | 24.00 ± 5.00ab | |
| EA | 46.00 ± 2.00b | 39.67 ± 2.52b | 50.67 ± 1.53b | 41.67 ± 2.52c | 44.00 ± 2.65b | 38.00 ± 2.00b | 50.33 ± 0.58b | 37.00 ± 2.00ab | |
| ME | 51.33 ± 2.52b | 47.33 ± 1.53c | 50.67 ± 1.53c | 48.33 ± 3.06c | 48.67 ± 2.08b | 45.00 ± 1.73c | 50.33 ± 0.58c | 45.33 ± 0.58b | |
| 20 | IF | 56.33 ± 6.66a | 42.00 ± 4.00a | 53.67 ± 8.02a | 49.00 ± 2.00a | 55.67 ± 6.81a | 41.00 ± 5.00a | 52.00 ± 7.55a | 45.00 ± 4.00a |
| HE | 59.33 ± 9.07a | 47.33 ± 2.56a | 57.00 ± 4.58a | 44.00 ± 4.00a | 57.00 ± 6.58a | 45.67 ± 3.51ab | 55.67 ± 4.73a | 40.00 ± 5.00a | |
| EA | 67.00 ± 3.00ab | 54.33 ± 2.52b | 71.00 ± 1.00ab | 56.00 ± 2.65a | 65.00 ± 2.65ab | 52.67 ± 3.51b | 69.67 ± 1.16ab | 52.00 ± 3.61a | |
| ME | 73.33 ± 2.52b | 67.00 ± 2.65c | 71.00 ± 1.00b | 68.67 ± 3.22a | 71.67 ± 2.08b | 66.00 ± 2.65c | 69.67 ± 1.16b | 65.33 ± 2.08b | |
| 30 |
IF | 69.67 ± 6.51a | 56.67 ± 4.16a | 65.67 ± 6.66a | 64.33 ± 3.51a | 68.67 ± 6.51a | 55.67 ± 3.21a | 64.33 ± 6.51a | 62.00 ± 4.36a |
| HE | 51.33 ± 38.42a | 63.33 ± 3.79ab | 68.00 ± 4.59a | 58.33 ± 4.04a | 71.67 ± 3.79a | 62.00 ± 4.00ab | 67.33 ± 4.04a | 55.00 ± 3.46a | |
| EA | 77.00 ± 1.00a | 68.33 ± 3.79b | 80.67 ± 2.52ab | 70.00 ± 4.36a | 75.67 ± 4.16a | 67.00 ± 4.36b | 79.67 ± 1.53ab | 64.67 ± 3.79a | |
| ME | 90.00 ± 1.73a | 76.00 ± 3.00c | 80.67 ± 2.52b | 78.00 ± 3.00b | 88.67 ± 2.52b | 76.00 ± 1.00c | 79.67 ± 1.53b | 75.00 ± 1.00b | |
| Negative Control |
DMSO +K2Cr2O7 |
8.00 ± 3.61 | 8.00 ± 2.00 | 8.00 ± 1.00 | 8.33 ± 0.58 | 5.33 ± 2.08 | 6.33 ± 1.53 | 6.67 ± 0.58 | 6.33 ± 0.58 |
| K2Cr2O7 | 10.33 ± 2.10 | 9.33 ± 1.53 | 10.33 ± 1.53 | 10.33 ± 0.58 | 8.67 ± 1.53 | 8.00 ± 1.73 | 8.33 ± 1.52 | 9.00 ± 1.00 | |
| Positive Control |
5% | 100.00 ± 0.00 | 100.00 ± 0.00 | 100 ± 0.00 | 100.00 ± 0.00 | 86.67 ± 10.69 | 86.67 ± 10.69 | 84.00 ± 1.00 | 82.00 ± 1.00 |
ME: Methanolic extract, HE: Hexane extract, EAE: Ethyl acetate extract, IF: Infusion extract, DMSO: Diméthylsulfoxide and K2Cr2O7: Potassium dichromate. The results are the mean ± SD of triplicate tests evaluated after 24 and 48 h of incubation at room temperature. For the same column same concentrations, values carrying the same superscript letter are not significantly different at p ≥ 0.05 (Student-Newman-Keuls test).
Table 1. Sporulation inhibition percentage of P. guajava extracts on different Eimeria strains.
In vitro anti-sporozoidal activities of P. guajava extracts:
Different concentrations of P. guajava extracts showed concentration dependent inhibition for viability of coccidial sporozoites of different Eimeria species as compared to control groups Control-I (DMSO) and Control-II (HBSS) as shown in (Table 2). According to our results, most extracts including aqueous extracts exhibited good antisporozoidal activities against E. flavescens, E. stiedae, E. intestinalis and E. magna strains at 1000 μg/ml. The highest viability inhibitory percentage was 97.00 ± 1.73% at a concentration of 1000 μg/ml of P. guajava methanolic extract against E. intestinalis strain (Table 2). The lowest efficacy was 8.67 ± 2.08% at a concentration of 125 μg/ml of the infusion extract against E. magna.
| Conc µg/ml | Extract | Incubation time and Eimeria strains | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 12 h | 24 h | ||||||||
| E. intestinalis | E. magna | E. flavescens | E. stedai | E. intestinalis | E. magna | E. flavescens | E. stedai | ||
| 125 | IF | 7.67 ± 3.06a | 15.00 ± 2.65a | 3.00 ± 4.36a | 13.00 ± 1.73a | 24.00 ± 11.53a | 8.67 ± 2.08a | 18.67 ± 3.06a | 24.67 ± 3.06a |
| HE | 9.33 ± 1.15a | 24.00 ± 3.00b | 5.67 ± 1.53a | 4.00 ± 3.61b | 20.33 ± 1.15ab | 9.33 ± 1.15a | 21.33 ± 2.08a | 16.00 ± 2.65b | |
| EA | 17.33 ± 3.06b | 26.00 ± 2.00b | 14.00 ± 4.36b | 15.33 ± 3.06b | 28.67 ± 2.52ab | 17.33 ± 3.06b | 29.67 ± 4.04b | 26.33 ± 1.53b | |
| ME | 20.67 ± 2.89b | 27.67 ± 1.53b | 21.67 ± 6.03b | 16.00 ± 1.00b | 38.33 ± 4.16b | 20.67 ± 2.89b | 38.00 ± 6.56c | 28.67 ± 1.53b | |
| 250 | IF | 9.33 ± 1.53a | 17.67 ± 2.52a | 8.00 ± 1.00a | 18.00 ± 2.65a | 22.00 ± 1.00a | 9.33 ± 1.53a | 24.33 ± 1.53a | 30.33 ± 3.21a |
| HE | 31.67 ± 3.21b | 29.33 ± 3.21b | 25.33 ± 6.66b | 6.00 ± 3.00b | 43.67 ± 3.21b | 31.67 ± 3.21b | 42.00 ± 6.56b | 18.67 ± 2.52b | |
| EA | 34.33 ± 3.51b | 34.33 ± 2.08b | 32.00 ± 3.61b | 22.33 ± 3.06b | 45.33 ± 4.04b | 34.33 ± 3.51b | 47.67 ± 4.51b | 35.33 ± 2.08b | |
| ME | 44.67 ± 8.50c | 44.33 ± 5.51c | 43.67 ± 6.66c | 33.33 ± 6.51c | 60.33 ± 6.11c | 44.67 ± 8.50c | 59.33 ± 6.35c | 44.67 ± 4.51c | |
| 500 | IF | 32.67 ± 4.51a | 32.00 ± 4.00a | 25.33 ± 2.08a | 31.67 ± 4.04a | 45.00 ± 4.00a | 32.67 ± 4.51a | 42.00 ± 2.00a | 43.00 ± 4.36a |
| HE | 42.67 ± 4.51b | 42.33 ± 4.16b | 35.33 ± 2.08b | 21.00 ± 5.00b | 54.00 ± 4.58b | 42.67 ± 4.51b | 51.67 ± 1.53b | 32.33 ± 3.06b | |
| EA | 41.00 ± 2.61b | 45.67 ± 2.52b | 38.33 ± 4.16b | 34.00 ± 2.00b | 53.00 ± 2.00b | 41.00 ± 2.65b | 55.00 ± 3.61b | 46.67 ± 2.52b | |
| ME | 45.67 ± 2.08b | 53.33 ± 1.53c | 46.33 ± 0.58c | 42.33 ± 0.58c | 58.33 ± 2.52b | 45.67 ± 2.08b | 61.67 ± 1.53c | 53.33 ± 3.06c | |
| 750 | IF | 52.67 ± 6.81a | 48.00 ± 4.00a | 48.00 ± 7.55a | 42.00 ± 4.00a | 63.33 ± 6.66a | 52.67 ± 6.81a | 64.67 ± 8.02a | 54.00 ± 2.00a |
| HE | 54.00 ± 6.56a | 53.33 ± 2.52a | 51.67 ± 4.73a | 37.00 ± 5.00ab | 66.33 ± 9.07a | 54.00 ± 6.56a | 68.00 ± 4.58a | 49.00 ± 4.00a | |
| EA | 62.00 ± 2.65ab | 60.33 ± 2.52b | 57.33 ± 1.53ab | 49.00 ± 3.61b | 74.00 ± 3.00ab | 62.00 ± 2.65ab | 74.33 ± 1.53ab | 61.00 ± 2.65b | |
| ME | 68.67 ± 2.08b | 73.00 ± 2.65c | 65.67 ± 1.15b | 62.33 ± 2.08c | 80.33 ± 2.52b | 68.67 ± 2.08b | 82.00 ± 1.00b | 73.67 ± 3.21c | |
| 1000 |
IF | 65.67 ± 6.51a | 62.67 ± 4.16a | 60.33 ± 6.51a | 59.00 ± 4.36a | 76.67 ± 6.51a | 65.67 ± 6.51a | 76.67 ± 6.66a | 69.33 ± 3.51a |
| HE | 68.67 ± 3.79a | 69.33 ± 3.79ab | 63.33 ± 4.04a | 52.00 ± 3.46b | 58.33 ± 38.42a | 68.67 ± 3.79a | 79.00 ± 4.58a | 63.33 ± 4.04ab | |
| EA | 72.67 ± 4.16a | 74.33 ± 3.79b | 69.33 ± 3.22ab | 61.67 ± 3.79b | 84.00 ± 1.00a | 72.67 ± 4.16a | 86.00 ± 3.00ab | 75.00 ± 4.36b | |
| ME | 85.67 ± 2.52b | 82.00 ± 3.00c | 75.67 ± 1.53b | 72.00 ± 1.00c | 97.00 ± 1.73a | 85.67 ± 2.52b | 91.67 ± 2.52b | 83.00 ± 3.00c | |
| Negative Control |
DMSO | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 |
| HBSS | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | 00 ± 00 | |
| Positive Control |
50µg /ml | 79.00 ± 1.00 | 83.67 ± 10.69 | 81.00 ± 1.00 | 78.00 ± 1.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 | 100.00 ± 0.00 |
ME: Methanol extract, HE: Hexane extract, EAE: Ethyl acetate extract, IF: Infusion extract DMSO: Diméthylsulfoxide, HBSS: Buffer Hanks buffered salt solution and K2Cr2O7: Potassium dichromate. The results are the mean ± SD of triplicate tests evaluated after 12 and 24 h of incubation at room temperature. For the same column same concentrations, values carrying the same superscript letter are not significantly different at p ≥ 0.05 (Student-Newman-Keuls test).
Table 2. Viability inhibitory percentage of P. guajava extracts on different Eimeria strains.
In vitro Antioxidant activities of P. guajava extracts
Effects of P. guajava extracts on the DPPH radical: The DPPH radical scavenging activity of different extracts of P. guajava was evaluated and the results are shown in (Table 3). All the extracts of P. guajava exhibited stronger antioxidant activities, compared to that of the standard antioxidant molecule (Vitamin C) used. The hot water extract showed the lowest activity at any concentrations with an inhibition percentage of 70.52% at 200 μg/ml, while the methanolic extract showed the highest activity (94.59%) at the concentration 200 μg/ml. However, there was no significant (p>0.05) difference between the activity of Vitamin C and that of the methanolic and ethylacetate extracts of P. guajava at the concentration 200 μg/ml.
| Extracts | Concentration of extract (µg/mL) and scavenging activity (%) | IC50 (µg/ml) | ||||
|---|---|---|---|---|---|---|
| 12.5 | 25 | 50 | 100 | 200 | ||
| IF | 42.074 ± 1.42bcd | 46.074 ± 0.33ab | 50.370 ± 0.78b | 55.555 ± 2.65b | 70,518 ± 1,96b | 102.831 ± 22.78ab |
| HE | 42.592 ± 3.17bcd | 47.037 ± 1.28ab | 56.666 ± 1.55c | 63.407 ± 4.20c | 86,296 ± 3,90d | 37.969 ± 13.59a |
| EA | 44.66 ± 1.99cd | 70.518 ± 2.11cd | 88.518 ± 2.21e | 90.296 ± 0.49e | 91,925 ± 0,61e | 2.879 ± 0.20a |
| ME | 47.185 ± 0.66d | 78.740 ± 4.25cd | 86.296 ± 4.10e | 92.074 ± 1.33e | 94,592 ± 0,32e | 2.168 ± 0.27a |
| Vitamin C | 76.178 ± 6.69e | 86.186 ± 0.62e | 87.262 ± 0.75e | 90.157 ± 1.03e | 93.465 ± 0.37e | 1,295 ± 0,14a |
For the same column, values carrying the same superscript letter are not significantly different at p ≥ 0.05 (Student-Newman-Keuls test). ME: Methanolic extract, HE: Hexane extract, EAE: Ethyl acetate extract, IF: Infusion extract.
Table 3. DPPH radical-scavenging activities of P. guajava.
The concentrations which inhibited 50% of DPPH (IC50) are presented in (Table 3). These results show that the hot water extract had a high IC50 (low activity). The ethyl acetate and the methanol extract of P. guajava had the lowest IC50 (i.e. had the highest activity). The methanol extract of P. guajava had the lowest IC50 (i.e. the highest activity).
Ferric reducing/antioxidant power (FRAP) of P. guajava extracts: The reducing power was determined by the Fe3+- Fe2+ transformation in the presence of the extracts of P. guajava, and the results obtained are shown in (Table 4). The hot water extract showed the lowest reducing power while the standard (Vitamin C) exhibited the highest reducing power at the concentrations of 100 and 200 μg/ml. At 100 μg/ml, there was no significant difference between the reducing power of Vitamin C (2,510 ± 0,65) and the methanolic extract of P. guajava (2,517 ± 0,01). However, the hot water extract showed the lowest optical densities (i.e. lowest reducing power) at every concentration. The remaining extracts exhibited varied activities from one extract to another at each concentration.
| Extracts | Concentrations (µg/ml) et absorbance (à 700 nm) | ||||
|---|---|---|---|---|---|
| 12.5 | 25 | 50 | 100 | 200 | |
| IF | 0.632 ± 0.08d | 0.642 ± 0.05d | 0.802 ± 0.07d | 0.999 ± 0.06ab | 1.285 ± 0.06b |
| HE | 0.783 ± 0.03e | 0.782 ± 0.03e | 0.940 ± 0.03d | 1.317 ± 0.03b | 1.691 ± 0.02c |
| EA | 0.625 ± 0.06d | 1.331 ± 0.04f | 1.354 ± 0.04f | 1.810 ± 0.02c | 2.317 ± 0.07e |
| ME | 1.691 ± 0.07g | 1.940 ± 0.03h | 2.31 ± 0.03h | 2.517 ± 0.05d | 2.908 ± 0.07g |
| Vitamin C | 0.028 ± 0.00a | 0.044 ± 0.00a | 0.056 ± 0.02a | 2.510 ± 0.65d | 6.339 ± 0.09h |
For the same column, values carrying the same superscripts letter are not significantly different at p ≥ 0.05 (Student-Newman-Keuls test). ME: Methanolic extract, HE: Hexane extract, EAE: Ethyl acetate extract, IF: Infusion extract.
Table 4. Ferric reducing power activities of P. guajava extracts.
Effects of P. guajava extracts on Nitric oxide: The results of the scavenging capacity against nitric oxide were recorded in terms of percentage inhibition as presented in (Table 5). The extracts of P. guajava showed considerable antioxidant potential. The methanolic and ethylacetate extracts revealed the highest percentage inhibition indicating the best nitric oxide scavenging activity. However, hexane extracts of P. guajava showed the lowest scavenging activity at every concentration.
| Extracts | Concentrations (µg/ml) et pourcentage d’inhibition (%) | ||||
|---|---|---|---|---|---|
| 12.5 | 25 | 50 | 100 | 200 | |
| IF | 86.295 ± 0.147a | 89.23 ± 0.327ab | 89.591 ± 0.269ab | 89.634 ± 0.374bc | 89.787 ± 0.274ab |
| HE | 81.029 ± 0.211a | 81.978 ± 2.037a | 84.003 ± 0.546ab | 84.349 ± 0.473b | 86.738 ± 3.725ab |
| EA | 83.271 ± 4.231b | 88.594 ± 0.725ab | 89.425 ± 0.798ab | 89.627 ± 0.385bc | 90.734 ± 0.672c |
| ME | 85.849 ± 1.725b | 86.257 ± 0.725c | 89.647 ± 0.258ab | 88.464 ± 11.151bc | 92.349 ± 0.729c |
| Vitamine C | 92.427 ± 3.627c | 94.595 ± 2.032c | 94.595 ± 1.339b | 96.556 ± 0.895c | 96.556 ± 0.298c |
| BHT | 94.946 ± 0.800c | 96.429 ± 0.110d | 97.274 ± 0.526c | 97.624 ± 0.027d | 99.410 ± 0.055d |
For the same column, values carrying the same superscript letter are not significantly different at p ≥ 0.05 (Student-Newman-Keuls test). ME: Methanolic extract, HE: Hexane extract, EAE: Ethyl acetate extract.
Table 5. Nitric oxide (NO) radical scavenging of P. guajava extracts.
Total phenolic content of P. guajava extracts: The total phenolic content of P. guajava extracts were determined in this study using Folin-Ciocateu Reagent method and the results are presented in (Table 6). The concentration of phenolic compounds in the methanolic extract (18,536 mgGAE/mg) was higher than in all other extracts. The methanolic and Ethyl Acetate had relatively the same concentration (p>0.05) and the lowest concentration of phenolic compounds was observed in the infusion extract (8.380 mgGAE/mg).
| Extracts | Phenols (mgGAE/mg) | Flavonoids (mgCE/mg) |
|---|---|---|
| Infusion | 8.380 ± 0.80bc | 0.494 ± 0.00ab |
| Hexane | 10.461 ± 1.20cd | 1.720 ± 0.13d |
| Ethyl Acetate | 15.328 ± 2.13ef | 1.881 ± 0.03d |
| Methanol | 18.536 ± 2.17f | 1.991 ± 0.18d |
Along each column, values with the same superscripts are not significantly different, Waller Duncan (P>0.05).
Table 6. Total phenolic and flavonoid contents of P. guajava extracts.
Total flavonoid content of P. guajava extracts: The total flavonoid contents of the various extracts are presented in (Table 6). The result obtained showed that the methanol extract had the highest flavonoid content (1,991 mgCE/mg) while the infusion extract showed the lowest value of flavonoid content.
Cytotoxicity test: In order to evaluate the cytotoxicity effect, L929, HEPG2 and Hella cells were exposed to P. guajava methanolic extract, for 48 h and cell grown inhibition was accessed using MTT assay. In our current study, the methanolic extract exhibited CC50 of >30 μg/ml against (Table 7) the selected cell lines, suggesting that the compounds are not toxic.
Selectivity index: The selectivity index of the methanolic extract was then evaluated using the MTT assay on L929, HEPG2 and Hella cells in order to check that their toxicity was specific to the parasite (Table 7). The impact of toxicity was established by analysing the selectivity index (SI) values. In our study, selectivity index values for the tested extract ranged between 1.01 to 20.64 μg/ml. The methanolic extract of P. guajava showed the highest selectivity index value of 20.64 μg/ ml, on L929 cells which was noteworthy as the extracts from this plant showed good anticoccidial activity.
| Plants | Cell line | CC50 (µg/ml) |
Sporozoidal IC50 (µg/ml) |
Selectivity index (µg/ml) |
|---|---|---|---|---|
| P. guajava | L929 cells | 148.83 | 94.99 | 20.64 |
| HEPG2 cells | 96.24 | 1.01 | ||
| Hella cells | 129.29 | 1.36 |
Table 7. Selectivity index, CC50 on L929, HEPG2 and Hella cells of P. guajava methanolic extracts.
Phytochemical analysis: Phytochemical screening of the most active extracts were consistent with detection of alkaloids, flavonoids, Saponines, Steroids and Tannins, whereas, the absence of polyphenols and terpenoids were noticed (Table 8).
| Chemical groups/Plant extract | P. guajava |
|---|---|
| Alkaloids | + |
| Flavonoids | + |
| Polyphenols | - |
| Tannins | + |
| Saponines | + |
| Steroids | + |
| Terpenoids | - |
Table 8. Phytochemical screening of P. guajava methanolic extracts.
Discussion
In Cameroon as in all developing countries, plants are regularly solicited by farmers to treat recurrent coccidioses. In this study, we evaluated the anticoccidial and antioxidant activities of crude extracts of one African traditional medicinal plant. The observations that P. guajava extract concentrations had an effect on the sporulation of coccidia oocysts indicates that P. guajava extracts are able to kill or inhibit growth and development of oocysts. The finding that P. guajava had the highest sporulation inhibition at 30 mg/ml suggests that it is more effective in treating coccidiosis. According to our results, most extracts including aqueous extracts exhibited good oocysticidal activity against Eimeria intestinalis, Eimeria magna, Eimeria flavescens and Eimeria stedai strains. The P. guajava extract showed maximum sporulation inhibition activity at 30 mg/ml and was observed to be more effective against Eimeria intestinalis. Similar to present findings, Molan et al. [32] also observed invitro sporulation inhibition with aqueous extracts of pine bark (Pinus radiata) in three species of avian coccidia. Since extracts have been shown to inhibit endogenous enzyme activities [33], then it is possible that P. guajava extract reduced the proportion of sporulation by inhibiting or inactivating the enzymes responsible for the sporulation process as in helminth eggs [34]. Jones et al. [34] suggested that extracts may penetrate the cell wall of oocysts and cause a loss of intracellular components. In the present study, the P. guajava extracts might have penetrated the wall of the oocysts and damaged the cytoplasm (sporont) as evidenced by the appearance of abnormal sporocysts in oocysts exposed to higher concentrations. The differences between the four extracts in inhibiting sporulation of coccidia oocysts may be due to differences in chemical composition. The observation that K2Cr2O7 could not inhibit sporulation could be explained by the fact that since it is a bactericidal drug as well, it might have killed the bacteria present thereby enhancing the sporulation of oocysts. Potassium dichromate killed bacteria in a sample containing coccidian oocysts thereby enhancing sporulation of coccidia oocysts. Therefore it could be that bacteria if present, could have interfered with the sporulation of oocysts, possibly by competing for nutrients and/or feeding on the oocysts.
The percentage of cells viability under control circumstances (DMSO and HBSS) in this study was comparable with other studies using Eimeria species [35,36], therefore the method used may be consi