Research Article - Biomedical Research (2017) Volume 28, Issue 4
Does the tumor necrosis factor a predictor factor in patients with chronic hepatitis B and C
Yilmaz Bilgic1*, Yuksel Seckin1, Yasir Furkan Cagin1, Oguzhan Yildirim1, Cengiz Yilmaz1, Murat Harputluoglu1, Hakan Harputluoglu2, Sami Akbulut3 and Abdurrahman Sahin41Department of Gastronterology, Faculty of Medicine, Inonu Unıversty, Turkey
2Department of Medical Oncology, Faculty of Medicine, Inonu Unıversty, Turkey
3Liver Transplant Institute, Faculty of Medicine, Inonu Unıversty, Turkey
4Department of Gastronterology, Faculty of Medicine, Fırat Unıversty, Turkey
- *Corresponding Author:
- Yilmaz Bilgic
Department of Gastronterology
Faculty of Medicine
Inonu Unıversty, Turkey
Accepted on September 14, 2016
Abstract
Aim: In our research, we aimed to investigate the importance and pattern of Tumor Necrosis Factor (TNF) alpha in response to the combinatorial treatment of lamuvidin (LAM) and Pegile interferon (PEG INF) 2a + Ribavirin (RIB) in patients with Chronic Hepatitis B (CHB) and Chronic Hepatitis C (CHC).
Material and Method: Thirty four CHB and 25 CHC patient samples were taken before and after the treatment and stored appropriately. CHB patients were treated with LAM and CHC patients were treated with using the combination of PEG INF 2a and RIB. HBV DNA, anti HBe, ALT, AST and TNF alpha results were obtained from CHB patients before and 48th week of the treatment. HCV RNA, ALT, AST and TNF alpha results were also obtained from CHC patients before and in the 6th month of completing the treatment. Responses to the treatment were taken as negative result of HBV DNA in CHB patients after finishing the treatment and as negative result of HCV RNA in CHC patients after 6 months of finishing the 48 weeks of PEG INF treatment.
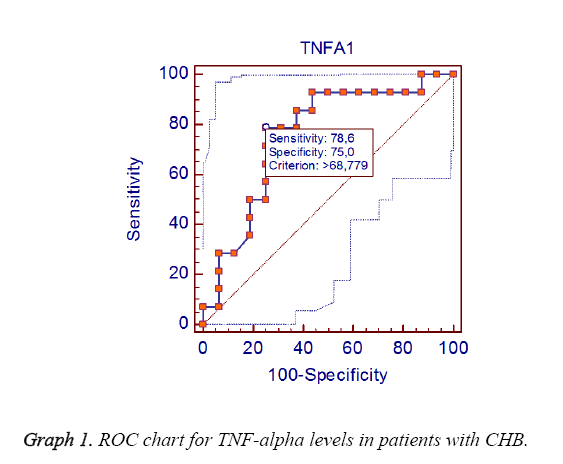
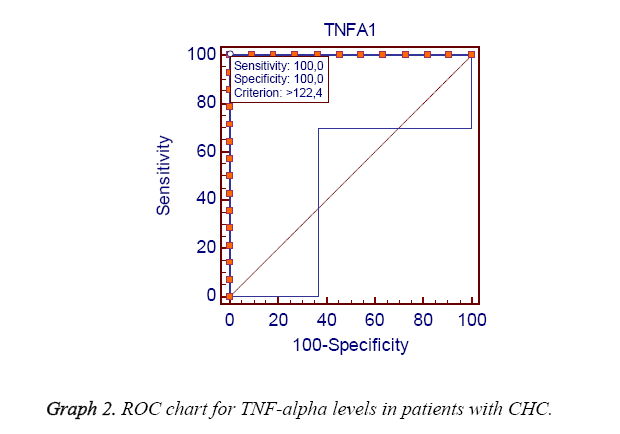
Results: At the level of decreasing TNF-alpha in CHB patients when the group responding to the treatment compared to the group not responding to the treatment, a significant difference was observed in favor of the respondent group (p<0.0001). At the level of decreasing TNF-alpha in CHC patients when the group responding to the treatment compared to the group not responding to the treatment, a significant difference was also obtained in favor of the respondent group (p<0.0001). In the analysis for emphasizing the importance of TNF alpha level in response to the treatment in CHB patients, it was found that predicting the response to the treatment of TNF alpha was 78.6% sensitivity and 75% specificity for 68.78 cutoff. In the analysis for emphasizing the importance of TNF alpha level in response to the treatment in CHC patients, it was found that determining the response of TNF alpha to the treatment was 100% sensitivity and 100% specificity for 122.4 cutoff.
Conclusions: With the treatment a decrease in TNF alpha is occured in CHB and CHC patients; however, TNF alpha decrease in the groups responding to the treatment is more significant. The results of this research show that TNF alpha level may be an important non-invasive marker for foresight and determining the response to the treatment.
Keywords
Hepatitis B, Hepatitis C, TNF alpha, Lamuvidin, Pegile interferon.
Introduction
CHB is still a common health problem on the world and is endemic in some areas. CHB may cause chronic hepatitis, cirrhosis and HCC. According to the host's immune response, HBV infection leads to different clinical outcomes. Pro-inflammatory and anti-inflammatory cytokines are responsible for the body's immune response and HBV clinical results occur depending on this response [1]. A number of factors such as host related factors (such as genetic and immunological factors), factors related to the virus (viral load, genotype etc.) and environmental factors (hygiene, nutritional status, treatment, vaccination, etc.) affect how to result of HBV infection [2]. In the immunopathogenesis of HBV infection, cytokines and regulatory molecules play a vital role gene polymorphism and gene loci of these cytokines and their molecules affect the HBV infection [3]. Gene polymorphisms of cytokines also affect the response to treatment and vaccination [4]. CHC is a major cause of chronic liver disease that affects 150-300 million people on the world [5]. Virus is eliminated 50% of patients who are infected with HCV using INF-based therapy [6]. There is a correlation between disease severity and TNF-alpha levels in HCV patients [7]. TNF-alpha is produced mainly in kupfer cells in HCV patients [8]. At CHC sustained virological response is defined as the negativity of HCV RNA in the 24th week after the treatment [9].
TNF-alpha is the key cytokine against host immune response and viral clearance of HBV. TNF alpha levels were increased in patients with HBV infection [10,11]. TNF-alpha is a cytokine produced mainly by activated macrophages [12]. TNF-alpha gene is localized in MHC HLA III region and levels of TNF alpha are affected by two gene polymorphisms in the promoter [13]. TNF-alpha is usually not detected in healthy individuals but in infections and inflammatory conditions serum and tissue levels of TNF-alpha are high [14]. TNF alpha serum levels are significantly correlated with the severity of the infection [15]. TNF-alpha in the liver has some functions like the two ends of a sharp sword. It is required for the proliferation of liver cells during liver damage. It is also a hepatotoxic mediator in many animal models. TNF-alpha is an important pathogenic mediator in patients with viral hepatitis [12].
Materials and Methods
In our study, patients with CHB and CHC were diagnosed by clinical, laboratory and liver biopsies in our clinic. Alcoholic liver diseases, autoimmune hepatitis, cholestatic hepatitis, primary biliary cirrhosis, Delta infections and unknown etiology of liver diseases were excluded from the research. 34 patients with CHB and 25 patients with CHC were included in the study. HBsAg antigen with patient serum samples of HBsAg (Austria II RIA kit, Abbott Laboratories, North Chicago, III, USA) and anti-HCV antibody with the serum samples of patients were evaluated with anti-HCV (secondgeneration enzyme-linked immunosorbent assay (ELISA); Abbot) kit. The viral nucleic acids were obtained before the liver biopsies. Serum HBV DNA was determined by southern blot hybridization method and HCV RNA (Amplicor HCV test, Roche Diagnostic System INC. Asia, Singapore) assay.
Thirty four CHB and 25 CHC patients samples were taken before and after the treatment and stored appropriately. CHB patients were treated with LAM and CHC patients were treated with using the combination of PEG INF 2a and RIB. HBV DNA, anti HBe, ALT, AST and TNF alpha results were obtained from CHB patients before and 48th week of the treatment. HCV RNA, ALT, AST and TNF alpha results were also obtained from CHC patients before and in the 6th month of completing the treatment. TNF-alpha was measured by ELISA. Responses to the treatment were taken as negative result of HBV DNA in CHB patients after finishing the treatment and as negative result of HCV RNA in CHC patients after 6 months of finishing the 48th week of PEG INF treatment.
Measuring of TNF-alpha
Five cc of blood samples were taken from patients and centrifuged at 4000 rpm for 5 minutes. Then, they were stored at -80°C. Frozen serum samples were melted in accordance with the procedure and resulting sera were studied with ELISA method using TNF-α kit (Biosource International, CA, USA).
Statistical analysis
Data were summarized by the mean and standard deviation or median with minimum and maximum values or number and percentage. The normal distribution suitability was assessed by Kolmogorov-Smirnov test. T test for dependent samples on statistical analysis, Wilcoxon paired two samples test and Mann-Whitney U test were used where appropriate. In order to determine the optimal cut-off point of TNF-alpha level in patients with CHB and CHC, receiver operating characteristic (ROC) curve technique was used. The values of P<0.05 were considered as statistically significant. IBM SPSS Statistics 23.0 software was used in the analysis.
Results
When patients with CHB and CHC were compared according to their genders, ages and post TNF alpha levels, there was not found a statistically significant relation (p>0.05) (Graphs 1 and 2). However, when patients were compared in terms of ALT, AST and pre TNF alpha levels, a significant difference was detected (p<0.01, 0.01, 0.001) as shown in Table 1. Decreasing of TNF-alpha level in patients with chronic hepatitis B had significant difference (p<0.0001) in favor of the respondent group when the group not responding to treatment was compared with the group responding to treatment. Moreover, decreasing of TNF-alpha level in patients with chronic hepatitis C had significant difference (p<0.0001) in favor of the respondent group when the group not responding to treatment was compared with the group responding to the treatment as shown in Table 2. In the analysis for emphasizing the importance of TNF alpha level in response to the treatment in CHB patients, it was found that predicting the response to the treatment of TNF alpha was 78.6% sensitivity and 75% specificity for 68.78 cutoff. In the analysis for emphasizing the importance of TNF alpha level in response to the treatment in CHC patients, it was found that determining the response of TNF alpha to the treatment was 100% sensitivity and 100% specificity for 122.4 cut-off.
| Variables | CHB (n=34) | CHC (n=25) | P values |
|---|---|---|---|
| Male (%) | 21(62) | 16 (64) | >0.05 |
| Female (%) | 13(38) | 9 (36) | >0.05 |
| Median years* | 36(18-55) | 32 (19-48) | >0.05 |
| ALT (IU/ml)** | 106.7 ± (37.4) | 68.16 ± (39.21) | <0.01 |
| AST (IU/ml)** | 74.94 ± (38.89) | 45.8 ± (20.59) | <0.01 |
| PreTTNFA**(pg/ml) | 122.19 ± (114.6) | 165.34 ± (111.08) | <0.001 |
| PostTTNFA**(pg/ml) | 56.5 ± (45.0) | 71.4 ± (29.9) | <0.05 |
| ALT: Alanine Transaminase; AST: Aspartate Transaminase; CHB: Chronic Hepatitis B; CHC: Chronic Hepatitis C; PreTTNFA: Pretreatment mean TNF alpha level; PostTTNFA: Post Treatment Mean TNF Alpha Level *The values in the brackets were expressed as minimum and maximum **The values in the brackets were expressed as standard deviation |
|||
Table 1. The demographic characteristics of the patients.
| CHB the patients (n=34) | HBV DNA after the treatment | P value | |
|---|---|---|---|
| Positive | Negative | ||
| Change in PostTTNFA*(pg/mL) | -18.5 ± (24.7) | -118 ± (89.3) | <0.0001 |
| CHC the patients (n=25) | HCV RNA after the treatment | ||
| Positive | Negative | ||
| Change in PostTTNFA*(pg/mL) | -26.8 ± (21.4) | -146.5 ± (83.7) | <0.0001 |
| CHB: Chronic Hepatitis B; CHC: Chronic Hepatitis C; TNF: Tumor Necrosis Factor; HBV DNA: Hepatitis B Virus Deoxyribo Nucleic Acid; HCV RNA: Hepatitis C Virus Ribo Nucleic Acid; PostTTNFA: Post Treatment Mean TNF Alpha Level *The values in the brackets were expressed as standard deviation |
|||
Table 2. Change in TNF alpha related with the HBV DNA and HCV RNA negativity after the treatment in CHB and CHC.
Discussion
TNF alpha levels had increased in both serum and liver tissues of the patients with CHB and CHC [16]. In the study to investigate the role of the TNF-alpha over the pathogenesis of CHB and CHC, both viruses had induced TNF-alpha expression in human liver cells [17]. Cytokines play an important role in starting and regulating of immune response; therefore, they affect the natural pattern and sensitivity of HBV infection [18]. In CHB, the secretion of TNF alpha and serum levels had increased in vitro. In IFN alpha therapy, TNF alpha level had significantly increased with transaminase elevations. Thus, it had suggested that this pattern showed the elimination of HBV [12]. When compared the changes in TNF-alpha level of CHB patients who responded and did not respond to treatment, change in the negative patient group was statistically significant (p<0.0001). In our study, TNF-alpha level was lowered by drug therapy in CHB patients but this low level occurred much more for patients who responded to the treatment. In the analysis for emphasizing the importance of TNF alpha level in response to the treatment in CHB patients, it was found that predicting the response to the treatment of TNF alpha was 78.6% sensitivity and 75% specificity for 68.78 cutoff.
TNF-alpha inhibits the replication and expression of HBV. Recombinant TNF alpha inhibits the replication of HBV after transplantation by increasing mRNA degradation of Hepatitis B Virus [19]. In the study, it had been shown that TNF alpha was higher than the control group. Moreover, it had been demonstrated that TNF alpha levels were decreased by being inhibited of HBV viral replication [12]. It had been suggested that TNF alpha were derived from mononuclear cells in the liver and in chronic liver disease TNF alpha might play a role in inflammatory activity [20]. TNF alpha is an important antiviral cytokine in the development of immune response at viral clearance against HBV [21]. The immune response against virus-infected liver cells is associated with liver damage. In the development of immune response, these cytokines have an important place [22]. In our study, a decrease in TNF alpha levels of CHB patients was observed. We suggest that the current treatment can be effective overimmune pathways which stimulate TNF alpha production. In addition, our research also supports that TNF alpha suppression which is a proinflammatory cytokine may be the immune-mediated response in the treatment of HBV.
The interferon treatment in CHC patients allows clearance of virus and reduces TNF-alpha levels in responding patients. Interestingly, pre-treatment levels of TNF alpha is higher than the patients who respond compared to the patients who do not respond [23]. It had been shown that in TNF alpha cell cultures, IFN signaling pathway inhibited HCV replication [24]. It had also been demonstrated that Etanerceptin, a TNF alpha inhibitor, contributed to the therapeutic effects of IFN alpha and ribavirin in HCV patients [25]. At the level of decreasing TNF-alpha in CHC patients when the group responding to the treatment compared to the group not responding to the treatment, a significant difference was also obtained in favor of the respondent group (p<0.0001). In the analysis for emphasizing the importance of TNF alpha level in response to the treatment in CHB patients, it was found that predicting the response to the treatment of TNF alpha was 78,6% sensitivity and 75% specificity for 68.78 cutoff. In the analysis for emphasizing the importance of TNF alpha level in response to the treatment in CHC patients, it was found that determining the response of TNF alpha to the treatment was 100% sensitivity and 100% specificity for 122.4 cutoff.
TNF-alpha is important for the initial immune response against Hepatitis C. With increasing of lymphocyte proliferation against HBV, CTL stimulation and thus the onset of antiviral activity had been shown [26]. The immune response against virus-infected liver cells is associated with liver damage. In the development of immune response these cytokines have an important place [22]. Applying of Peg-IFN allows the secretion from soluble TNF-alpha receptors. Thus, it has been argued that TNF mediated immune response is inhibited by Peg-IFN therapy [27]. It had been suggested that TNF alpha and interferon gamma (IFN-g) had important roles involving the stimulation of cytotoxic T cells and the activation of natural killer cells for the development of the host immune response [28]. We conclude that decreasing of TNF alpha levels with peg interferon treatment suppresses HCV virus with the treatment, and ultimately the suppression of TNF alpha stimulating pathways may be occured. Moreover, with peg interferon-2a treatment TNF alpha suppression, which is the inflammatory cytokine, supports that immune-mediated response may be in HCV treatment.
Natural Killer cells inhibit viral replication without causing any damage to liver cells through increasing the production of TNF alpha and IFN gamma [29,30]. Macrophages through Toll-like receptor expression mainly increase the production of cytokines such as interleukin (IL) 21b, IL-6 and TNF alpha [31]. In the phase II study, it had been reported that Etanerceptin INF, a TNF-alpha antibody, in combination with ribavirin significantly reduced HCV replication (For the etanercept treatment group HCV RNA reduction was 63%; on the other hand, for IFN and ribavirin treatment group it was 32%.) [25]. It had been suggested that in de novo infections TNF alpha increased the input of HCV by disrupting the development of the innate immune response in liver cells [32,33]. In our study, a decrease in TNF alpha level was observed with Pen INF 2a and RIP treatments. We suggest that the current treatment can be effective over immune pathways which stimulate TNF alpha production. In addition, our research also supports that TNF alpha suppression which is a proinflammatory cytokine may play a role in the pathogenesis of CHC and be the immune-mediated response in the treatment of HCV.
Conclusions
It is shown that TNF alpha be an important non-invasive marker for foresight and determining the response to the treatment both CHB and CHC patients. In addition, it can also be said that TNF alpha may be used in the management of new patients with CHB and CHC.
References
- Tunçbilek S. Relationship between cytokine gene polymorphisms and chronic hepatitis B virus infection. World J Gastroenterol 2014; 20: 6226-6235.
- Thursz M. Genetic susceptibility in infectious diseases. Biotechnol Genet Eng Rev 2000; 17: 253-264.
- Thursz MR. Host genetic factors influencing the outcome of hepatitis. J Viral Hepat 1997; 4: 215-220.
- McNicholl JM, Downer MV, Udhayakumar V, Alper CA, Swerdlow DL. Host-pathogen interactions in emerging and re-emerging infectious diseases: a genomic perspective of tuberculosis, malaria, human immunodeficiency virus infection, hepatitis B, and cholera. Annu Rev Public Health 2000; 21: 15-46.
- http://ecdc.europa.eu/en/healthtopics/hepatitis_c/pages/index.aspx
- Pawlotsky JM, Chevaliez S, McHutchison JG. The hepatitis C virus life cycle as a target for new antiviral therapies. Gastroenterol 2007; 132: 1979-1998.
- Kallinowski B, Haseroth K, Marinos G, Hanck C, Stremmel W, Theilmann L. Induction of tumour necrosis factor (TNF) receptor type p55 and p75 in patients with chronic hepatitis C virus (HCV) infection. Clin Exp Immunol 1998; 111: 269-277.
- Zylberberg H, Rimaniol AC, Pol S. Soluble tumor necrosis factor receptors in chronic hepatitis C: a correlation with histological fibrosis and activity. J Hepatol 1999; 30: 185-191.
- European Association for Study of Liver. EASL Clinical Practice Guidelines: management of hepatitis C virus infection. J Hepatol 2014; 60: 392-420.
- Sheron N, Lau J, Daniels H, Goka J, Eddleston A, Alexander GJ, Williams R. Increased production of tumour necrosis factor alpha in chronic hepatitis B virus infection. J Hepatol 1991; 12: 241-245.
- Zhang G, Li Z, Han Q, Li N, Zhu Q, Li F, Lv Y, Chen J, Lou S, Liu Z. Altered TNF-α and IFN-γ levels associated with PD1 but not TNFA polymorphisms in patients with chronic HBV infection. Infect Genet Evol 2011; 11: 1624-1630.
- Bradham CA, Plümpe J, Manns MP, Brenner DA, Trautwein C. Mechanisms of hepatic toxicity. I. TNF-induced liver injury. Am J Physiol 1998; 275: G387-392.
- Abraham LJ, Kroeger KM. Impact of the-308 TNF promoter polymorphism on the transcriptional regulation of the TNF gene: relevance to disease. J Leukoc Biol 1999; 66: 562-566.
- Nurnberger W, Platonov A, Stannigel H, Beloborodov VB, Michelmann I, von Kries R. Definition of a new score for severity of generalized Neisseria meningitidis infection. Eur J Pediatr 1995; 154: 896-900.
- Waage A, Halstensen A, Espevik T. Association between tumour necrosis factor in serum and fatal outcome in patients with meningococcal disease. Lancet 1987; 1: 355-357.
- Fang JW, Shen WW, Meager A, Lau JY. Activation of the tumor necrosis factor-alpha system in the liver in chronic hepatitis B virus infection. Am J Gastroenterol 1996; 91:748-753.
- Gonzalez-Amaro R, Garcia-Monzon C, Garcia-Buey L, Moreno-Otero R, Lonso JL, Yague E, Pivel JP, Lopez-Cabrera M, Fernandez-Ruiz E, Sanchez-Madrid F. Induction of tumor necrosis factor α production by human hepatocytes in chronic viral hepatitis. J Exp Med 1994; 179: 841-848.
- Grünhage F, Nattermann J. Viral hepatitis: human genes that limit infection. Best Pract Res Clin Gastroenterol 2010; 24: 709-723.
- Tsui LV, Guidotti LG, Ishikawa T, Chisari FV. Posttranscriptional clearance of hepatitis B virus RNA by cytotoxic T lymphocyte- activated hepatotytes. Proc Natl Acad Sci USA 1995; 92: 12398-12402.
- Yoshioko K, Kakumu S, Arao M, Tsutsumi Y, Inoue M, Wakita T. Immunohistochemical studies of intrahepatic tumor necrosis factor alpha in chronic liver disease. J Clin Pathol 1990; 43: 298-302.
- Jung MC. Pape GR, immunology of hepatitis B infection. Lancet Infect Dis 2002; 2: 43-50.
- Lewinson W, Jawetz E. Immunology. In: Du¨ ndar IH, Transl. ed. Medical Microbiology and Immunology. Istanbul: Barıxs Kitabevi/Appleton and Lange, 1998, 327-400.
- Larrea E, Garcia N, Qian C, Civeira MP, Prieto J. Tumor necrosis factor a gene expression and the response to interferon in chronic hepatitis C. Hepatology 1996; 23: 210-217.
- Lee J, Tian Y, Chan ST, Kim JY, Cho C, Ou JH. TNF-α Induced by Hepatitis C Virus via TLR7 and TLR8 in Hepatocytes Supports Interferon Signaling via an Autocrine Mechanism. PLoS Pathog 2015; 11: e1004937.
- Zein NN, Etanercept Study G. Etanercept as an adjuvant to interferon and ribavirin in treatment-naive patients with chronic hepatitis C virus infection: a phase 2 randomized, double-blind, placebo-controlled study. J Hepatol 2005; 42: 315-322.
- Kim DW, Suziki R, Harada T, Saito I, Miyamura T. Transsuppression of gene expression by hepatitis C viral core protein. Jpn J Med Sci Biol 1994; 47: 211-220.
- Neuman MG, Benhamou JP, Ibrahim A, Malkiewicz I, Spircu T, Martinot-Peignoux M, Shear NH, Katz GG, Akremi R, Bourliere M, Marcellin P. Role of cytokines in the assessment of the severity of chronic hepatitis C and the prediction of response to therapy. Rom J Gastroenterol 2002; 11: 97-103.
- Neuman M. Cytokines and inflamed liver. Clin Invest Med 1998; 23: 1-6.
- Edwards-Smith CJ, Jonsson JR, Purdie DM, Bansal A, Shorthouse C, Powell EE. Interleukin-10 promoter polymorphism predicts initial response of chronic hepatitis C to interferon alfa. Hepatology 1999; 30: 526-530.
- Makowska Z, Duong FHT, Trincucci G, Tough DF, Heim MH. Interferon- and interferon- signaling is not affected by interferon-induced refractoriness to interferon-in vivo. Hepatology 2011; 53: 1154-1163.
- Thomas E, Gonzalez VD, Li Q. HCVinfection induces a unique hepatic innate immune response associated with robust production of type III interferons. Gastroenterol 2012; 142: 978-988.
- Liaskou E, Wilson DV, Oo YH. Innate immune cells in liver inflammation. Mediat Inflamm 2012.
- Fletcher NF, Sutaria R, Jo J, Barnes A, Blahova M, Meredith LW, Cosset FL, Curbishley SM, Adams DH, Bertoletti A, McKeating JA. Activated macrophages promote hepatitis C virus entry in a tumor necrosis factor-dependent manner. Hepatology 2014; 59: 1320-1330.