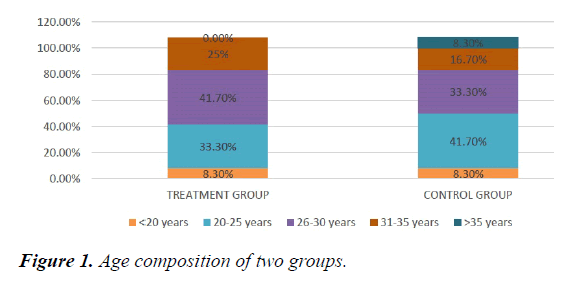Research Article - Journal of Pharmacology and Therapeutic Research (2018) Volume 2, Issue 2
Dengue and doxycycline-experience in a tertiary care hospital in eastern India in the year 2017 - An initial report.
Boudhayan Bhattacharjee*, Subhayan Bhattacharya, Bikash Sardar, Pushkar Raj, Moumita Ghosh, Debajyoti Majumdar, Arindam Naskar, Manab Kumar Ghosh, Sudeshna Mallik and Bibhuti Saha
Department of Tropical Medicine, School of Tropical Medicine, Kolkata, India
- *Corresponding Author:
- Dr. Boudhayan Bhattacharjee
Department of Tropical Medicine
School of Tropical Medicine
108, Chittaranjan Avenue, Kolkata
West Bengal, Pin 700073, India
Tel: +91-9477031946
E-mail: bou.bhatta@gmail.com
Accepted on June 06, 2018
Citation: Bhattacharjee B, Bhattacharya S, Sardar B, et al. Dengue and doxycycline-experience in a tertiary care hospital in eastern India in the year 2017 - An initial report. J Pharmacol Ther Res 2018;2(2):14-17.
DOI: 10.35841/pharmacology.2.2.14-17
Visit for more related articles at Journal of Pharmacology and Therapeutic ResearchAbstract
Dengue fever is an arthropod-borne viral haemorrhagic fever. Suspected dengue patients admitted in the of School of Tropical Medicine, Kolkata were taken as a case after considering inclusion and exclusion criteria. Aim of this study was to determine the effect of doxycycline in course and final outcome of dengue fever. Selected patients were subjected to history, examination, necessary investigations and then were managed according to NATIONAL VECTOR BORNE DISEASE CONTROL PROGRAMME (NVBDCP) guidelines. A total of 24 serology proven dengue patients were included and followed up. Two groups (twelve in each) were allocated by simple random sampling. Treatment group was given doxycycline and control group were given placebo. Age and sex profile were similar in both groups. 75% of patients in treatment group had no bleeding with no patients having major bleed; where in control group 25% of had major bleeding. Plasma leakage were less in treatment group (25%) as compared to control group (58.3%). Hemodynamic compromise was more in control group (16.7%) as compared to treatment group (8.30%). 41.70% of patients in treatment group had no thrombocytopenia in comparison to control group (16.70%). Also, only 8.30% of patients had platelets below 10,000/cmm in treatment group where it is 16.70% in control group. Platelet transfusion was necessary in 8.30% of treatment group patient in comparison to 16.70% of control group patients. Liver involvement in control group was more in comparison to treatment group. Control group has pancreatic involvement in 16.60% of patients; renal involvement in 8.30% of patients; cardiac involvement in 8.30% of patients. Whereas no pancreatic, renal or cardiac involvement were seen in treatment group. An initial report of this study showed significant reduction in complications of dengue fever in respect to different organ involvement when treated with doxycycline.
Keywords
Haemorrhagic fever, Dengue, Doxycycline, Thrombocytopenia
Introduction
Dengue fever is an arthropod-borne viral hemorrhagic fever caused by arbo-virus of flavivirus genus having 4 serotypes and concerned vectors like Aedes aegypti and Aedes albopictus mosquitoes. A clinical manifestation ranges from self-limiting fever to profound hemorrhage, shock and death. Dengue fever is described clinically as an acute febrile illness of 2-7 days duration with two or more of the following symptoms headache, rash, retro-orbital pain, myalgia, arthralgia, hemorrhagic tendencies [1]. Dengue hemorrhagic fever is diagnosed in a case with clinical dengue fever with hemorrhagic tendencies manifested by one or more of the following, a) positive tourniquet test b) petechiae, purpura or ecchymoses c) bleeding from mucosa, injection sites, gastrointestinal tract, or other sites and thrombocytopenia (<100 000 cells/cmm) and plasma leakage due to increased vascular permeability, manifested by one or more of the following: 1) A rise in hematocrit for sex and age ≥ 20%, 2) >20% drop in hematocrit after volume replacement treatment compared to baseline, 3) plasma leakage (pleural effusion, ascites, hypoproteinemia) [1]. Dengue shock syndrome is DHF and evidence of circulatory failure with rapid and weak pulse and narrow pulse pressure (<20 mmHg) or hypotension for age, cold skin or restlessness [1]. Four serotypes of Dengue (1,2,3 and 4) have been found from India [1]. In West Bengal dengue fever occurs mainly in rainy season from July to November and causes threat to people every year in West Bengal [2]. Several studies have been done to evaluate effects of doxycycline on course and outcome of dengue fever. Meta-analysis of Jenny Lyn Mangulabnan et al. [3] showed that doxycycline helped to lower the serum IL-6 and serum TNF in Day 3 and Day 7 post treatment in dengue fever patients. Zavala Castro et al. [4] concluded that Doxycycline treated patients show a higher immune regulation of cytokines IL-6, IL-1, and TNF, with an increase in the levels of IL-1ra and sTNF-R1, followed by the tetracycline treated groups which presents a slightly higher regulation than the control group without treatment. Terry M et al. [5] suggested that doxycycline can be beneficial to dengue patients at high risk of complications; mediated by reducing pro-inflammatory cytokines. Rothan HA et al. [6] showed inhibitory effect of doxycycline against dengue virus (higher activity against DENV2 and DENV4 as compared to DENV1 and DENV3). Castro JE et al. [7] depicted as doxycycline to be more effective than tetracycline in modulating cytokine and cytokine receptors. Yang J-M et al. [8] found inhibitory effects of tetracycline derivatives in flavivirus group (DENV 2). Bethell DB [9] studied that higher cytokine level in dengue patient associated with more complication. Aim of our study was to determine the treatment effect of doxycycline in dengue fever patients.
Aims and Objectives
To evaluate effect of doxycycline in course and outcome of dengue fever.
Materials and Methods
Study area
The patients admitted to the inpatient department (IPD) of Tropical Medicine department in School of Tropical Medicine, Kolkata. They have come from different areas of whole West Bengal and adjacent states.
Study population
Diagnosed as dengue patients (adults and children) who were admitted at the to the inpatient department (IPD) Tropical Medicine department of School of Tropical Medicine, Kolkata.
Study design
This was an observational and interventional study.
Study duration
From July 2017 to December, 2017.
Inclusion criteria
Serology confirmed dengue patients (Dengue NS1Ag or Dengue IgM or both; by ELISA method) and who wished to participate in the study.
Exclusion criteria
Unwilling patients
Patients with associated infection (bacterial/viral/parasitic)
Patients with co- morbidities (heart diseases, chronic liver disease, chronic kidney disease etc.)
Patients admitted with complications of dengue (like thrombocytopenia, hemorrhage, shock, plasma leakage etc.)
Sample size
24 (twenty-four)
Study Technique
Suspected dengue patients admitted in the IPD were taken as case after considering inclusion and exclusion criteria. After history and clinical examination blood samples on the first day of were drawn for routine complete hemogram, renal function tests, liver function test, lipid profile and were sent to Department of Biochemistry and Laboratory Medicine, STM for testing. Complete hemogram was done on daily. X Ray chest and ultra-sonography of whole abdomen, Electrocardiogram were done to exclude complications like pleural effusion, ascites or GB edema and any co-morbidities. Other investigations were done as per clinical condition during treatment.
Patients were randomly arranged in two groups. One group (treatment group) were given tab Doxycycline 100 mg twice daily for 5 days other group (control group) were given tab placebo. Patients were managed and followed up as per NATIONAL VECTOR BORNE DISEASE CONTROL PROGRAM (NVBDCP) guideline. Treatment received and outcome (in respect to complications and mortality) were recorded. Collected data were tabulated and analyzed according to standard statistical methods.
Tools of Study
Pre-designed proforma with history, clinical examination and blood reports with dengue viral serology
Automated analyzer for biochemical and pathological tests
Machine for Dengue Viral serology (by ELISA)
X ray chest machine
Ultra-sonography machine
ECG machine
Statistical Analysis
Collected data were analyzed using appropriate statistical methods.
Ethics
The study protocol was reviewed and approved by the Clinical Research Ethics Committee of Calcutta School of Tropical Medicine, Kolkata.
Patient Information and Informed Consent
According to recent ICMR Guidelines informed consents were taken.
Results
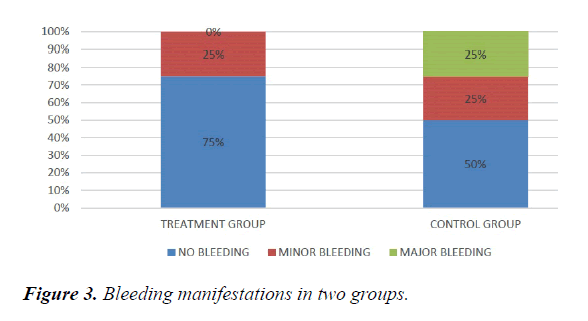
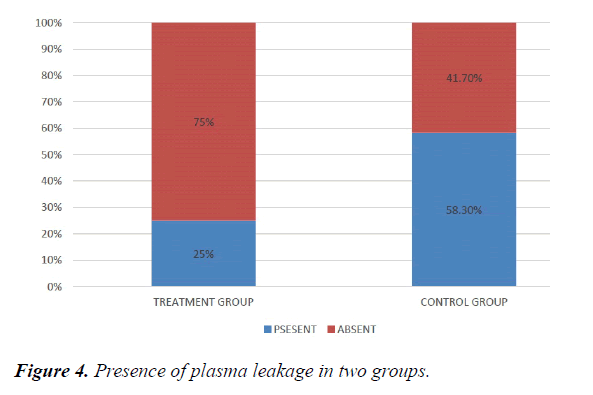
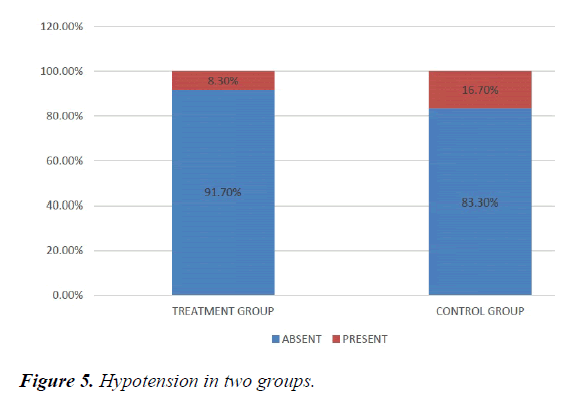
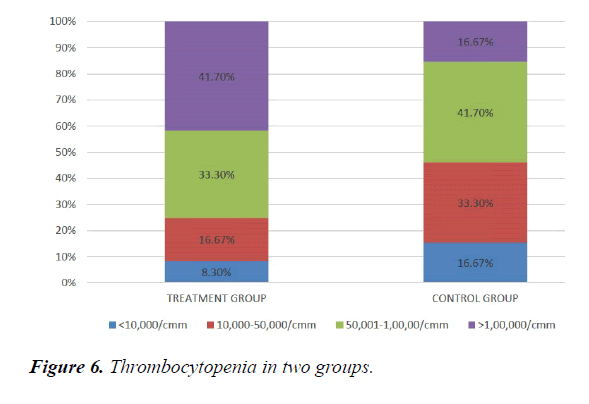
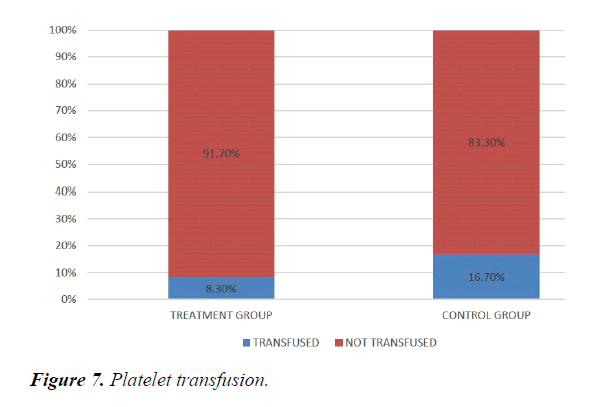
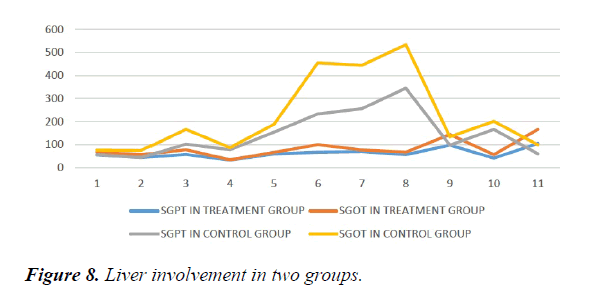
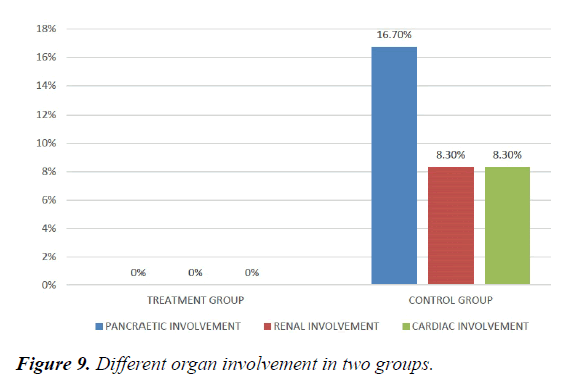
We studied total 24 (twenty-four) serologically proven dengue patients. Age and sex composition in both groups were similar (Figures 1 and 2). Treatment group has significantly less bleeding manifestations in comparison with control group (Figure 3). 75% of patients in treatment group had no bleeding with no patients having major bleed; where in control group 25% of had major bleeding (Figure 3). Plasma leakage (in the form of ascites /pleural effusion on both) were less in treatment group (25%) as compared to control group (58.3%) (Figure 4). Hemodynamic compromise was more in control group (16.7%) as compared to treatment group (8.30%) (Figure 5). 41.70% of patients in treatment group had no thrombocytopenia in comparison to control group (16.70%) (Figure 6). Also, only 8.30% of patients had platelets below 10,000/cmm in treatment group where it is 16.70% in control group (Figure 6). Platelet transfusion was needed in 8.30% of treatment group patient in comparison to 16.70% of control group patients (Figure 7). Liver involvement in control group was more in comparison to treatment group in respect to both SGOT and SGPT elevation (Figure 8). Control group has pancreatic involvement (raised amylase/ lipase or both) in 16.60% of patients; renal involvement (raised urea/creatinine) in 8.30% of patients; cardiac involvement (myocarditis/arrhythmias) in 8.30% of patients (Figure 9). Whereas no pancreatic, renal or cardiac involvement were seen in treatment group (Figure 9).
Discussion
Meta-analysis of Jenny Lyn Mangulabnan et al. [3] showed that doxycycline was favoured in lowering the serum IL-6 and serum TNF, both in Day 3 and Day 7 post treatment in patients with dengue fever. But this meta-analysis did not show specific organ involvement data. In our study age and sex composition in both treatment and control groups were similar. Treatment group has significantly less bleeding manifestations in comparison with control group; where in control group 1/4th of had major bleeding. Plasma leakage was significantly less in treatment group (more than 50% of control group).
Hemodynamic compromise was twice in control group as compared to treatment group. Thrombocytopenia was about 1/3rd less in treatment group Also, platelets below 10,000/cmm twice in control group in comparison with treatment group. Platelet transfusion was required twice in percentage in control group. Liver involvement in control group was more in comparison to treatment group. Control group has pancreatic involvement (raised amylase/ lipase or both) in 16.60% of patients; renal involvement (raised urea/creatinine) in 8.30% of patients; cardiac involvement (myocarditis/arrhythmias) in 8.30% of patients. Whereas no pancreatic, renal or cardiac involvement were seen in treatment group.
Conclusion
All over world different clinical manifestations of the dengue fever in different epidemics have been observed. This study showed significant reduction in complications of dengue fever when treated with doxycycline in respect to different organ involvement. However, this just an initial report of our study showing promising results with doxycycline. In future this study will yield more data regarding efficacy of doxycycline in dengue fever when we can analyze data on larger number of patients.
References
- National Guidelines for Clinical Management of Dengue Fever, National Vector Bourne
Disease Control Programme. 2015. - “Glimpses of IDSP works West Bengal 2012”. Disease surveillance Down to Earth
Edition 2013. Vol II. - Mangulabnan JL, Ogbac F. Effects of doxycycline on lowering IL-6 and TNF among patients with dengue haemorrhagic fever: a meta-analysis. 27th ESCMID. 2017.
- Zavala Castro JE, Fredeking TM. Doxycycline modify the cytokine storm in patients with
dengue and dengue haemorrhagic fever. Int J Infect Dis. 2010;14(S1):e44. - Fredeking TM, Zavala-Castro JE, González-Martínez P, et al. Dengue Patients Treated with Doxycycline showed lower mortality associated to a reduction in IL-6 and TNF levels. Recent Pat Antiinfect Drug Discov. 2015;10(1):51-58.
- Rothan HA, Mohamed Z, Paydar M, et al. Inhibitory effect of doxycycline against dengue virus replication in vitro. Arch Virol. 2014;159(4):711-718.
- Castro JE, Vado-Solis I, Perez-Osorio C, et al. Modulation of cytokine and cytokine receptor/antagonist by treatment with doxycycline and tetracycline in patients with dengue fever. Clin Dev Immunol. 2011;2011:1-5.
- Yang J-M, Chen Y-F, Tu Y-Y, et al. Combinatorial computational approaches to identify tetracycline derivatives as Flavivirus inhibitors. PLoS One. 2007;2(5):e428-441.
- Bethell DB, Flobbe K, Cao XT, et al. Pathophysiologic and prognostic role of cytokines in dengue hemorrhagic fever. J Infect Dis. 1998;177(3):778-782.